
Essential medical supplies are in high demand in today’s current environment, including surgical gloves. One way to fulfill this demand is to import gloves from foreign countries. When importing medical devices of any kind, including something as simple as gloves, you’ll still need to consider government regulations, customs clearance, tariffs, and more.
Depending on product details and the country of origin, duty rates on nitrile gloves imported into the U.S. range from zero to 14% of the shipment’s total value. It’s important to use the correct HTS code when you import these goods. Otherwise, you risk being fined by CBP due to product misclassification incorrect import duties.
Nitrile gloves are part of a larger group of medical gloves that includes examination gloves and special chemotherapy gloves designed specifically for handling chemotherapy products. These are considered personal protective equipment, according to the U.S. Food and Drug Administration (FDA).
They aid healthcare workers and patients by protecting their hands from infectious germs or harmful substances.
Single-use gloves such as those used in the medical field are mostly classified under Harmonized System code 4015 — articles of apparel & access of unhardened rubber. According to data from trade.gov, this code accounted for approximately $2.2 billion worth of goods imported to the USA in 2023, with the following three countries taking up the lion’s share of that business.
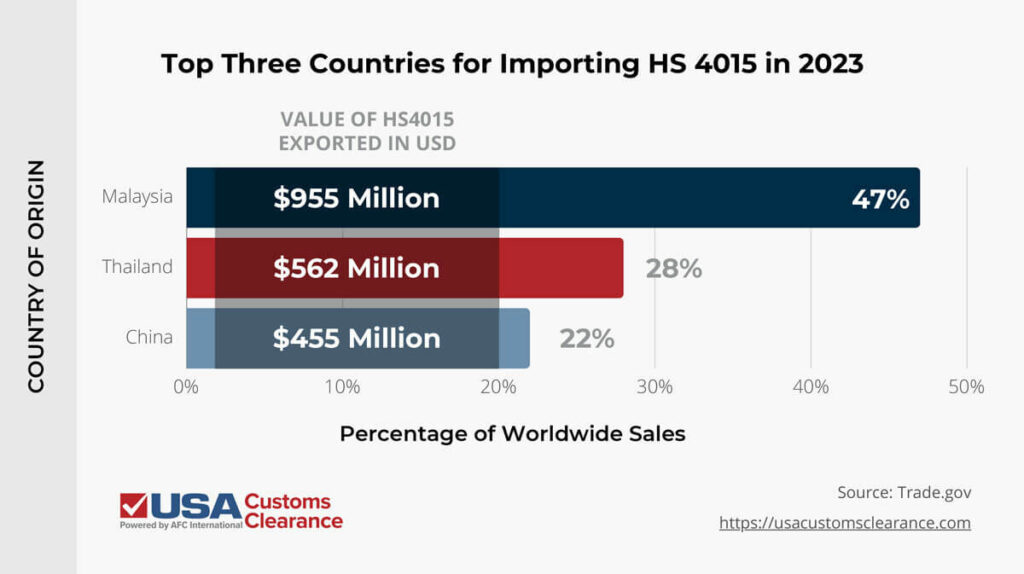
While China once dominated this market, changes to trade policies have made sourcing Chinese goods less appealing for importers in the United States.
In 2018, the U.S. government began applying additional tariffs on nearly all goods imported from China. These new restrictions significantly impacted trade between the two world powers.
In May 2024, the Biden Administration announced a new set of tariffs on medical glove imports from China, increasing the rate of duty from 7.5% to 25%, starting in 2026.
Surgical gloves are grouped together with other types of medical gloves by the FDA. These items range in design from basic gloves used for general examinations to specialized versions used to handle hazardous materials.
They’re further classified as personal protective equipment (PPE). This large classification encompasses a variety of products that are meant to prevent the spread of infection, illness, and injury.
Other examples of PPE include:
If your goods meet FDA standards, you can initiate the import process. Many importers choose to speak with an import consultant at this point. Working with a consultant reduces the likelihood that difficulties will arise during customs clearance. This includes complete loss of goods due to seizure by CBP, which many importers experienced during the pandemic in 2020.
If you’re unsure about the import compliance of your shipment, we recommend working with one of our experienced customs brokers to avoid delays, fines, and penalties.
Gloves intended for medical use, including surgery, are categorized as class one medical devices. The FDA reviews these goods to ensure that they meet specific criteria related to safety and performance. Their regulations may require you to file a 510(k) premarket notification prior to bringing such goods into the U.S.
For gloves that have already been approved by the FDA, you’ll simply need to reference the existing approval rather than filing a new one. If items have been significantly modified since their initial approval, you will probably need to submit a new 510k form for the agency to review.
In 2017, the FDA banned the use of powder as a lubricant in single-use medical gloves, including those used for patient examinations and surgical applications. This ban applies to disposable gloves of all materials and extends to veterinary use.
The ban is limited to clinical purposes, so powdered gloves can still be used in lab research, janitorial tasks, food preparation, and other situations that don’t involved contact with a patient. However, they still have the potential to cause swelling and inflammation to the wearer after extended use.
If you plan on importing these goods for medical use in the USA, it’s important to make sure they’re not of the powdered variety.
One of the most important aspects of the import process is ensuring you have a customs bond when necessary. A bond is required when importing $2,500 or more worth of goods. This is also required when the goods in question are regulated by government agencies. Therefore, you’ll need a bond to import gloves for medical use.
 Copy URL to Clipboard
Copy URL to Clipboard
Did you find this article helpful?
See more of our coverage on Google.
Add usacustomsclearance.com as a preferred source!

Licensed customs support for importers across a wide range of U.S. entry needs. USA Customs Clearance provides Customs Bonds, Consulting, Customs Brokerage, Manifest Confidentiality, Importer of Record support, and Guides & Resources to help importers prepare for U.S. Customs and Border Protection (CBP) requirements and customs clearance with regulatory compliance, greater clarity, and confidence.
With licensed broker support, transparent service information, and secure checkout, we help importers take the right next step.
I want to import Nitrile Exam Gloves .
I want to know if i require 510k . What are the current tariffs to obtain 510k if i require it.
Hello, i am in China,if we don't have FDA 510K, my customer who want to buy nitrile glove for food use, what's the duty rate?
Hey there,
I’m shipping a container of exam gloves to the US and we’re told that we need a device license number which I am having a really hard time attaining. I’m addition, POA docs have been signed, we’re bonded, trucking company has been hired but I’m worried that the goods will not clear customs. What are our options?
Hi Franky,
One of our import experts will reach out to you shortly to provide assistance with your situation. We look forward to helping you!
What is The tariff for powder free nitrile exam gloves. And is the LST number required to import these?
Hi Wilkin,
The tariff amount for powder free nitrile gloves can vary based on the country of origin. An LST number isn't always required. However, at least one Affirmation of Compliance code will be required at the time of entry and an LST is often used for this purpose.
Our Licensed Customs Brokers can assist you with both of these tasks. You can schedule a consulting session with our brokers at the link below:
Customs Broker Consulting
i am going to import nitrile gloves from China to UsA. The manufacturer needs a 510k? Also the importer or buyer needs a 510K? who needs it.
Hi Kevin,
A 510K is generally needed for medical devices that are marketed and sold in the U.S. However, due to the current situation in regards to the global pandemic the FDA has made some temporary exceptions to this rule. We've responded privately with additional information. We look forward to working with you!
Hi Sir/ Madam,
Good Day to you!
I am in the midst of exporting 15k boxes (1.5k carton) Nitrile Examination Gloves to America, the product consist of FDA approval. I just want to ask the below two questions:
a) Document needed such as Country of Origin (COO)?
b) The product box behind never state the manufacturer but it did state the endorsement by the US & EU Representative namely (removed for privacy), Germany respectively. Of course, we have the sufficient and legit documents to prove this product is from this manufacturer. With the above mentioned, is it still able to import to America?
Kindly clarify and sincerely thank you for the respond!
Hi Cyril,
We've responded privately with the information you requested. We look forward to assisting you.
Can importer use ASTM cert instead of FDA cert to import Nitrile gloves into US?
Hi Adil,
The CBP and FDA are handling some of these excpetions on a case by case basis due to the immense need for medical supplies including nitrile gloves. We strongly recommend consulting with a Licensed Customs Broker before proceeding with importing your gloves.
Consult with our Licensed Customs Brokers to get the help you need. We look forward to assisting you.
Greetings.
I am reseller for NITRILE GLOVES from a Thailand manufacturer. I have buyers who want to buy and export to Los Angeles a sum of 20million boxes of Nitrile gloves. I would like to know the certifications and the procedure required to export the above from Thailand to Los Angeles or any state in the US.
I thank you for your information.
Hi Uma,
You'll need to comply with all standard customs requirements including covering your shipment with a customs bond. Additionally, depending on the intended use of the gloves (medical vs. non-medical) you'll need to provide additional documentation and ensure the proper labeling guidelines are followed.
We've reached out privately with additional information that will assist you further. We look forward to working with you.
Dear Sir /Madam,
I am looking to import nitrile gloves and other medical items from China - what duty is applicable for gloves and PPE in general from China to the USA? Do I have to register my company in the USA as an importer of PPE? If so, how would I do so?
Kind regards,
Mark
Hi Mark,
The import duty for your items will depend on the specific HTS code that's applicable for your products. One of our Licensed Customs Brokers can assist you with this. We've responded privately with the informaionyou requested related to becoming a registered importer also. We look forward to assisting you!
Gentlemen,
I would be importing about 200,000 boxes of Nitrile medical exam gloves from Vietnam or probably Thailand and I would like to contract your custom clearance services. Can you please tell me the rates I would need to pay for your fees once the good arrive in Los Angeles and the corresponding tarrif I would need to pay (if any).
Also, I would like to know if non-medial face masks such as kn95 pay any import duties when coming from China.
Look forward to your prompt answer and thanks so much for your prompt answer.
Hello Mario,
In order to provide you with the tariff information that you're requesting, one of our Licensed Customs Brokers will need to properly classify the HTS code of the products that you're looking to import.
We've responded privately to your message with this information, as well additional details that you requested. We look forward to working with you!
I want to import Nitrile Exam Gloves FDA registered under 510k Exempt. What are the current tariffs?
Hi Tarek,
We've privately with the information that you requested. We look forward to working with you!
Hi,could you please show a sample of fda 510k cert which requires by custom?
Hi Grace,
We've reached out privately to provide you with this information. We look forward to working with you!
Some people told me Disposable Nitrile Gloves from Vglove and Top Glove and are banned recently by US Customs. Is it true?
Hi Tom,
Gloves manufactured by WRP Asia Pacific Sdn. Bhd were banned from being imported in September of 2019. However, this ban was lifted on March 23, 2020. VGlove and Top Glove haven't had any bans recently and are currently allowed to be imported.
If you're looking to import nitrile gloves and have further questions or need assistance, our Licensed Customs Brokers are available to help you. You can reach out to them at (855)912-0406 or schedule a consulting session today. We look forward to assisting you.
I am looking to import into USA - General purpose hospital use NITRILE GLOVES from Malaysia & Vietnam.Kindly advise me is there any import Duty on this item and what are the other official requirements to import this item.Thanks.
Hi Zameer,
In most cases, there is no duty rate for this product. However, depending on the size, material construction, and country of origin a duty rate may apply and will typically range from 3-4%. If your gloves are intended for hospital use, they'll need to be in compliance with all FDA requirements.
Our Licensed Customs Brokers can further advise on your upcoming imports and ensure that everything is properly in place to avoid unnecessary charges or delay of your goods. You can schedule a customs consulting session with our Licensed Customs Brokers to get answers to all of your importing questions. We look forward to helping you, Zameer.